Any content you receive is for information purposes only. Always conduct your own research.*Sponsored
See Why Glucotrack, Inc. (Nasdaq: GCTK) Will be Topping the Krypton Street Watchlist Tomorrow Morning—Tuesday, April 21, 2026 Don’t Miss The Next Breakout—Get Real-Time Alerts Sent Directly To Your Phone. Up To 10X Faster Than Email.
***Expect Full Coverage On (GCTK) To Begin Early***
Take A Look At (GCTK) Before Tomorrow Morning…
April 20, 2026
Now on Deck | Get (Nasdaq: GCTK) On Your Radar Before Tomorrow Morning
Dear Reader,
After today’s low-float AI profile made an approximate 80% move, we’re already turning our attention to what just hit our radar for tomorrow.
And this next story starts with something surprisingly simple:
A small patch.
Worn on the arm. Replaced every couple of weeks.
For many living with diabetes, that routine has become part of daily life.
But Glucotrack, Inc. (Nasdaq: GCTK) is working on something very different: a fully implantable continuous blood glucose monitor designed to track glucose levels continuously for years—with no on-body wearable component at all.
No patch.
No replacements every 10–14 days.
No visible device.
And with early human data already in hand and a key FDA filing expected in the coming weeks, (GCTK) is starting to draw attention as it moves into a pivotal stage of development.
Let me show you what’s going on.
Glucotrack, Inc. (Nasdaq: GCTK) is developing a fully implantable continuous blood glucose monitor designed for long-term use—without the on-body wearable component required by today’s standard CGM systems.
Which is exactly why (GCTK) will be topping our watchlist tomorrow morning—Tuesday, April 21, 2026.
Right now, (GCTK) appears to be flying under the radar, currently trending below $1, as of this week.
But keep in mind, (GCTK) has less than 1.8M shares listed as available to the public. When companies have small public floats like this, the potential exists for big moves if demand begins to shift.
Recently, (GCTK) made an approximate 53% move in less than a week, from around $.65 on April 9 to $1.00 on April 13. 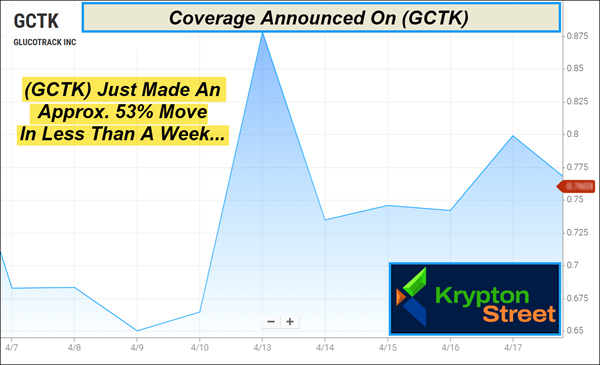
That combination—a differentiated device, early human data, and a development timeline that is beginning to tighten—is starting to put (GCTK) in a very interesting position for us. In a space dominated by wearable systems, a company working on a fully implantable alternative is naturally going to stand out. And when that story is paired with a small public float and a recent move that already showed this name can react sharply, it becomes easier to see why more eyes could begin shifting here. In other words, the setup is no longer just conceptual—it is starting to look like a story the market may have to pay closer attention to. With a pivotal FDA filing weeks away and a U.S. clinical trial slated for the second half of 2026, (GCTK) is quickly becoming one of the most intriguing names in the medtech diabetes space.
To understand why this story is starting to stand out, it helps to take a closer look at what (GCTK) is building—and why its approach could be so different from what patients use today.
Company Overview
Glucotrack, Inc. (Nasdaq: GCTK) is a Rutherford, New Jersey–based medical technology company focused on the design, development, and commercialization of novel technologies for people with diabetes.
The company's flagship program is its Continuous Blood Glucose Monitor (CBGM)—a long-term implantable system that measures blood glucose from blood, not interstitial fluid, eliminating the calibration lag that plagues traditional wearable CGM devices.
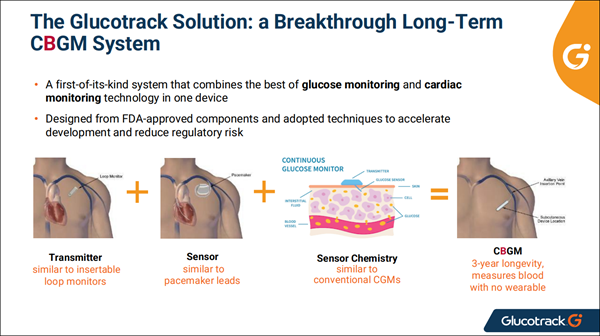
The CBGM is designed for a 3-year sensor longevity, requires minimal calibration, and has no on-body wearable component—a major quality-of-life boost for many of the people currently wrestling with wearable patches, adhesives, and frequent sensor replacements.
Unlike first-generation subcutaneous sensors that max out at 14 days, or larger pacemaker-class devices, (GCTK)’s design bridges a clear gap in the market: long-term, low-burden, fully implanted glucose visibility.
The company has already completed a first-in-human study in Brazil and a follow-up feasibility study in Australia, generated three issued U.S. patents, secured a U.S. clinical trial site, and engaged a CRO—all culminating in an IDE submission to the FDA planned for Q2 2026.
The Market Potential : Diabetes Is a Massive and
Growing Problem

The scale of the diabetes epidemic is staggering.
The International Diabetes Federation reports approximately 589M adults worldwide are currently living with diabetes—a figure expected to reach 700M by 2045.
Diabetes caused at least $1T in health expenditure – a 338% increase over the last 17 years. Against that backdrop, the global CGM market was valued at approximately $13.3B in 2025 and is projected to grow to $31.4B by 2031 at a 15.4% CAGR, making it one of the fastest-growing segments in all of medical devices.
Today's market is dominated by wearable subcutaneous sensors from Abbott, Dexcom, and Medtronic—three companies that collectively controlled over 98% of 2025 shipments according to Mordor Intelligence.
But every one of their devices requires periodic on-body replacement.
(GCTK) is targeting the patient population that wants continuous monitoring without the daily or biweekly maintenance burden.
That represents a genuinely differentiated niche within a rapidly expanding market.
Clinical Progress & Regulatory Readiness
(GCTK)’s development program is now at a critical inflection point.
The company completed a first-in-human study in São Paulo, Brazil between December 2024 and January 2025.
The 5-day in-hospital study enrolled 10 participants with Type 1 or Type 2 diabetes on intensive insulin therapy. Results were compelling: a Mean Absolute Relative Difference (MARD) of 7.7% across 122 matched pairs, a 99% data capture rate, and zero procedure- or device-related serious adverse events.
The study met all primary and secondary endpoints.
In July 2025, the company initiated a multicenter feasibility study in Australia at St. Vincent's Hospital in Melbourne.
Learnings from that study have been incorporated into product design iterations ahead of the U.S. program.
A U.S. clinical site has been secured, a CRO has been engaged, and the company confirmed it expects to file its Investigational Device Exemption (IDE) with the FDA in Q2 2026, targeting a U.S. trial launch in the second half of 2026, subject to FDA approval.
Intellectual Property & Financial Position
In October and November 2025, the USPTO issued three critical patents for (GCTK)'s CBGM platform (Patent Nos. US 12,453,494, US 12,458,257, and US 12,458,258), covering proprietary sensor chemistry, intravascular lead design, and low-power electronics.
These patents protect the core innovations that differentiate (GCTK)'s system from both short-lived subcutaneous sensors and larger, more invasive implantable devices.
On the financial side, (GCTK) reported $7.4M in ca-sh and ca-sh equivalents as of December 31, 2025, up from $5.6M at the prior year-end, aided by a $4.0M private placement completed in December 2025.
Full-year 2025 R&D expenses were $9.8M, reflecting continued investment in product design, manufacturing, and preclinical work.
The company stated that existing cash is sufficient to fund its 2026 operating plan through at least the end of Spring 2026—covering the IDE submission.
Management has assembled a leadership team with deep backgrounds at Dexcom, Abbott, Senseonics, and Medtronic.
7 Reasons Why GCTK Will Be Topping Our Watchlist Tomorrow Morning—Tuesday, April 21, 2026…
1. Implantable Edge: Glucotrack (Nasdaq: GCTK) is developing a fully implantable continuous blood glucose monitor designed for years of use with no on-body wearable component, setting it apart from standard CGM systems.
2. Tight Float: With less than 1.8M shares listed as available to the public, (GCTK)’s small public float could have the potential for big moves if demand begins to shift.
3. Recent Momentum: (GCTK) recently made an approximate 53% move in less than a week, rising from around $.65 on April 9 to $1.00 on April 13.
4. Human Data: Early clinical results for (GCTK) showed a 7.7% MARD across 122 matched pairs, a 99% data capture rate, and zero procedure- or device-related serious adverse events.
5. FDA Timing: The company said (GCTK) expects an IDE submission to the FDA in Q2 2026, with a U.S. clinical trial targeted for the second half of 2026, giving the story a defined near-term timeline.
6. Patent Protection: Three issued U.S. patents give (GCTK) protection around key parts of its platform, including sensor chemistry, intravascular lead design, and low-power electronics.
7. Market Potential: (GCTK) is going after a differentiated segment within a global CGM market projected in the report to grow from about $13.3B in 2025 to $31.4B by 2031, focused on people who want continuous monitoring without frequent wearable replacement.
Take A Look At (GCTK) Before Tomorrow Morning…

When you step back and look at what’s coming together here, the picture starts to get very interesting.
Glucotrack, Inc. (Nasdaq: GCTK) is developing a fully implantable continuous blood glucose monitor designed for long-term use, without the on-body wearable component required by today’s standard systems.
That alone makes (GCTK) stand out.
And the setup around this one is hard to ignore.
With fewer than 1.8M shares available to the public, (GCTK)’s small float could witness the potential for big moves if demand begins to shift.
It recently just made an approximate 53% move from around $.65 to $1.00 in less than a week.
There’s also real progress behind the story. Early clinical data showed a 7.7% MARD across 122 matched pairs, a 99% data capture rate, and zero procedure- or device-related serious adverse events.
Now add a defined timeline, with an IDE submission expected in Q2 2026 and a U.S. clinical trial targeted for the second half of 2026, along with three issued U.S. patents protecting key parts of the platform.
Put it all together, and (GCTK) is becoming a name that deserves a closer look.
We will have all eyes on (GCTK) tomorrow morning.
Take a look at (GCTK) before you call it a night.
And watch for my next update, it could be coming bright and early.
Sincerely,
Alex Ramsay
Co-Founder / Managing Editor
Krypton Street Newsletter
|





No comments:
Post a Comment