Any content you receive is for information purposes only. Always conduct your own research. |
*Sponsored |
Daily Edge Report Just Put bioAffinity Technologies, Inc. (Nasdaq: BIAF) On Today's Watchlist—Tuesday, March 10, 2026 |
Don't miss the next breakout — get real-time alerts sent straight to your phone! |
Full Coverage on (BIAF) Begins Now
Take A Look At (BIAF) While It's Still Early… |
March 10, 2026 |
First Look | See Why (BIAF) is Getting Early Coverage This Morning |
Dear Reader, |
Early cancer detection is one of the most important frontiers in modern healthcare.
The earlier physicians can determine whether a suspicious finding is truly malignant, the greater the potential impact on both patient outcomes and clinical decision-making. |
Nowhere is that challenge more visible than in lung cancer screening. |
Each year, millions of patients undergo scans that reveal pulmonary nodules, small spots in the lungs that can indicate a wide range of possibilities. In many cases, physicians must rely on repeat imaging, biopsies, or other invasive procedures before determining whether cancer is actually present. A significant percentage of those procedures ultimately confirm benign results, highlighting the need for better front-line diagnostic tools. |
As a result, healthcare providers and researchers are increasingly turning toward noninvasive diagnostic technologies designed to help evaluate cancer risk earlier and more precisely. This shift is helping drive rapid innovation across the lung cancer diagnostics landscape, a market projected to reach $10.4B by 2034. |
One company positioning itself within this transition is bioAffinity Technologies, Inc. (Nasdaq: BIAF). |
(Nasdaq: BIAF) is advancing a diagnostic platform built around flow cytometry and proprietary cellular analysis, designed to help physicians evaluate lung nodules using noninvasive sample testing. By applying advanced laboratory techniques to detect early signals associated with malignancy, the company aims to provide clinicians with additional data to guide patient management. |
This evolving approach to lung cancer diagnostics is one reason (BIAF) is topping our watchlist this morning — Tuesday, March 10, 2026. |
There is also a structural element worth noting. |
According to MarketWatch, (BIAF) reports fewer than 5M shares listed as available to the public, creating a relatively tight public float. In smaller-cap healthcare names, limited share availability can sometimes lead to increased volatility if activity begins to shift. |
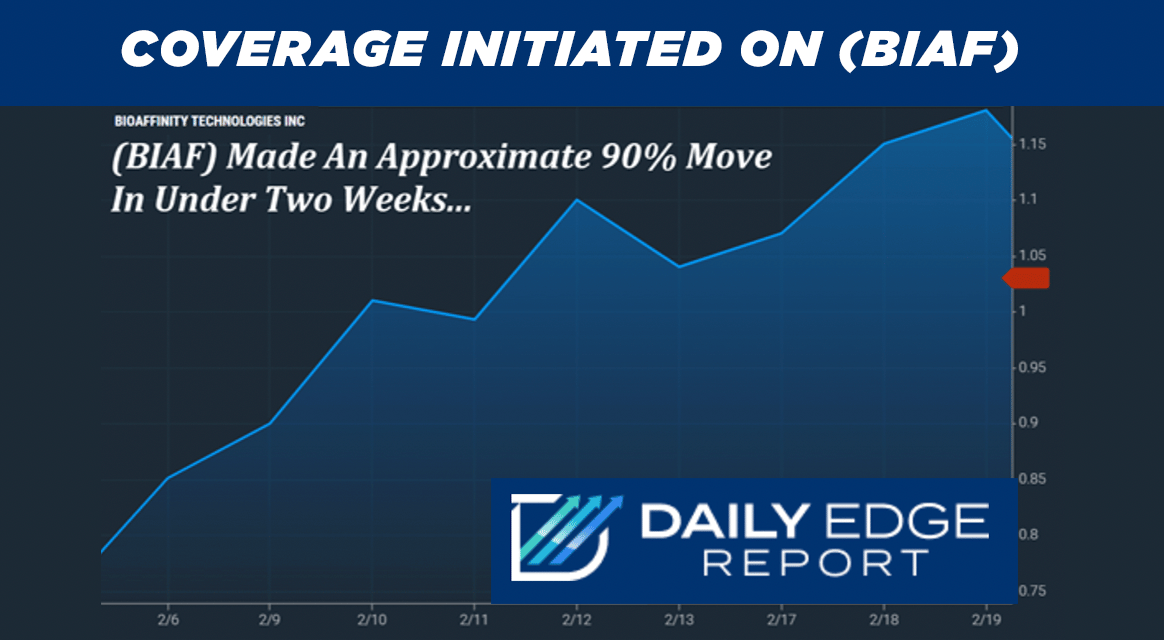
Recent action has already reflected periods of heightened activity.
Data shows that (BIAF) moved roughly 90% in under two weeks, climbing from approximately $0.73 on February 6 to $1.39 on February 19. |
Meanwhile, the company continues advancing a diagnostic alternative aimed at improving clinical decision-making when lung nodules are detected, an area where traditional approaches often lead to invasive procedures that later prove unnecessary. |
And as we began digging deeper into the company's technology, market positioning, and clinical approach, it became clear why (BIAF) is beginning to attract attention. |
|
Inside the Technology Driving Early Lung Cancer Detection |
bioAffinity Technologies (Nasdaq: BIAF) is a biotechnology company focused on developing noninvasive diagnostics and targeted technologies designed to detect cancer and other lung diseases earlier and more accurately. (BIAF)'s work centers on analyzing the cellular environment of the lungs to identify biological signals associated with disease, helping physicians make more informed decisions when evaluating patients with suspicious findings on imaging scans. |
At the core of (BIAF)'s platform is CyPath® Lung, the company's flagship diagnostic product. CyPath® Lung is a noninvasive test designed to help detect early-stage lung cancer, particularly in patients whose low-dose CT scans reveal pulmonary nodules that require further evaluation. |
The test works by analyzing sputum samples using advanced flow cytometry combined with proprietary analysis and artificial intelligence, allowing clinicians to identify cell populations that may indicate malignancy within the lung microenvironment. |
In clinical studies involving high-risk patients with small lung nodules, CyPath® Lung demonstrated 92% sensitivity and 87% specificity, highlighting its potential to support earlier detection while helping reduce unnecessary invasive procedures such as biopsies. |
CyPath® Lung is commercialized through Precision Pathology Laboratory Services, a bioAffinity subsidiary that processes samples and delivers physician reports based on the company's proprietary analysis platform. |
Beyond its flagship diagnostic, (BIAF) is also advancing broader research into cell-analysis technologies aimed at identifying multiple lung diseases and improving early cancer detection, leveraging flow cytometry and molecular analysis to characterize the lung microenvironment at the cellular level. |
Together, these technologies position (BIAF) within the growing field of precision diagnostics, where noninvasive testing tools are increasingly being used to help physicians detect disease earlier and guide patient care more effectively. |
|
A $10.4B Diagnostic Market |
The U.S. lung cancer diagnostics sector is expected to expand significantly over the coming decade, with projections indicating the market could grow to approximately $10.4B by 2034. A major driver behind this expansion is the increasing focus on detecting lung cancer earlier, particularly as the disease continues to rank among the leading causes of cancer-related mortality worldwide. |
Current screening protocols recommend annual low-dose CT (LDCT) scans for millions of individuals considered at elevated risk. While these scans play an important role in identifying suspicious lung nodules, they frequently leave physicians facing a difficult question: whether the finding represents cancer or a benign condition. This uncertainty can often lead to additional imaging, ongoing monitoring, or invasive procedures. |
bioAffinity Technologies, Inc. (Nasdaq: BIAF) is working to address this diagnostic gap with its CyPath® Lung test, which is designed to provide physicians with additional clinical insight after a suspicious lung nodule has been identified. |
In recently discussed case material, the test helped support a watchful surveillance approach for an elderly patient whose imaging had revealed a concerning nodule. Follow-up imaging later showed the nodule had resolved, reinforcing the decision to avoid a more invasive and costly biopsy. |
As healthcare providers continue searching for tools that improve diagnostic clarity, the ability to help rule out malignancy with greater confidence may represent a meaningful advantage in a field where false-positive findings can frequently lead to unnecessary medical intervention. |
Recent Developments Worth Watching (Nasdaq: BIAF) |
Several recent announcements have helped bring bioAffinity Technologies (Nasdaq: BIAF) into clearer focus as the company advances its noninvasive lung cancer diagnostic platform and expands clinical visibility. |
Research Presented to the American Cancer Society Lung Cancer Roundtable: The company reported that its CyPath® Lung test would be highlighted at the American Cancer Society National Lung Cancer Roundtable, where clinical case studies demonstrated the test's ability to identify Stage 1A lung cancer in diagnostically complex patients. These findings illustrated how the technology may help clinicians navigate uncertain lung nodule cases and potentially accelerate diagnosis. |
Validation Study: (BIAF) announced a validation study with Brooke Army Medical Center aimed at expanding sample collection options for the CyPath® Lung test. The collaboration is intended to support broader clinical use of the company's noninvasive diagnostic platform and improve access to testing. |
New Case Studies Demonstrating Clinical Utility: Recent case studies released by the company highlighted real-world examples where CyPath® Lung helped clarify lung nodule diagnoses, demonstrating its potential role in reducing diagnostic uncertainty for patients undergoing lung cancer screening. |
Intellectual Property Expansion for Lung Cancer Detection Technology:
(BIAF) also reported that the Australian Patent Office accepted a patent application covering the CyPath® Lung diagnostic method, strengthening bioAffinity's intellectual property portfolio for detecting early-stage lung cancer using automated flow cytometry and machine learning. Once issued, the patent is expected to extend protection for the technology through 2042. |
Capital Raise to Support Commercial Growth: In October 2025, (BIAF) completed a $1.8M registered direct financing, selling 720,000 shares at $2.50 per share. (BIAF) stated that the proceeds would be used to support working capital and help expand sales of the CyPath® Lung diagnostic test. |
Revenue Growth Driven by Increasing Test Adoption: (BIAF)'s financial results for the third quarter of 2025 showcased a strategic pivot. While total revenues were $1.4M, the company reported a 122% year-over-year increase in CyPath® Lung testing revenue. This growth was driven by higher case volumes and increased adoption within the Veterans Administration (VA) health system. |
|
7 Factors Putting (BIAF) On Our Radar This Morning — Tuesday, March 10, 2026 |
Limited Float: Fewer than 5M shares listed as available to the public, creating a relatively tight share structure.
Recent Activity: Barchart data shows an approximate 90% move in under two weeks, from about $0.73 on Feb. 6 to $1.39 on Feb. 19.
Expanding Market: The U.S. lung cancer diagnostics market is projected to reach $10.4B by 2034.
Noninvasive Approach: The company's CyPath® Lung test analyzes sputum samples to help evaluate potential lung malignancy.
Clinical Data: Reported study results showed 92% sensitivity and 87% specificity in nodules under 2 cm.
Technology Platform: Research presented at the AAAAI Annual Meeting highlighted potential applications in analyzing immune receptor responses related to asthma therapies.
Business Growth: CyPath® Lung testing revenue increased 122% year-over-year, with $7.7M in cash reported at the end of Q3 2025.
|
Take A Look At (BIAF) While It's Still Early… |
(BIAF) recently moved onto our radar for several reasons. The company reports fewer than 5M shares listed as available to the public, creating a relatively tight share structure that can sometimes lead to increased volatility when activity shifts. |
Recent chart data also shows notable activity, with (BIAF) making an approximate 90% move in under two weeks earlier this month. |
At the same time, the company is operating within the lung cancer diagnostics market, a sector industry projections suggest could grow to $10.4B by 2034. |
Beyond the market potential, the company's CyPath® Lung test is designed as a noninvasive sputum-based diagnostic tool intended to help physicians evaluate suspicious lung nodules before moving directly to invasive procedures. Clinical data cited for the test reports 92% sensitivity and 87% specificity when identifying cancer in nodules smaller than 2 centimeters. |
The underlying platform may also have broader potential. Research presented earlier this month suggested the technology could help identify antibody receptor responses linked to certain asthma therapies. |
Operational progress is beginning to appear in the company's financials as well. CyPath® Lung testing revenue increased 122% year-over-year, and the company reported $7.7M in cash at the end of Q3 2025. |
We have all eyes on (BIAF) this morning. |
Take a look at (BIAF) while it's still early. |
Also, keep a lookout for our next update, it could be on its way to you before the bell rings. |
Sincerely, |
Joel Locke |
Senior Editor |
The Daily Edge Report |
DailyEdgeReport.com ("DailyEdgeReport" or "DER" ) is owned by GG Media Holdings LLC, a single member limited liability company. Data is provided from third-party sources and DER is not responsible for its accuracy. Make sure to always do your own research and due diligence on any day and swing profile DER brings to your attention. Any emojis used do not have a specific defined meaning, and may be used inconsistently. We do not provide personalized in-vest-ment advice, are not in-vest-ment advisors, and any profiles we mention are not suitable for all in-vest-ors. |
Pursuant to an agreement between GG Media Holdings LLC and TD Media LLC, GG Media Holdings LLC has been hired for a period beginning on 03/09/2026 and ending on 03/10/2026 to publicly disseminate information about (BIAF:US) via digital communications. Under this agreement, TD Media LLC has paid GG Media Holdings LLC seven thousand five hundred USD ("Funds"). These Funds were part of the seventy seven thousand five hundred USD funds that TD Media LLC received from a third party named Sica Media LLC who did not receive the Funds directly or indirectly from the Issuer and does not own stock in the Issuer but the reader should assume that the clients of the third party own shares in the Issuer, which they will liquidate at or near the time you receive this communication and has the potential to hurt share prices. |
Neither GG Media Holdings LLC, TD Media LLC and their member own shares of (BIAF:US). |
Please see important disclosure information here: https://dailyedgereport.com/disclosure/biaf-2Xr7H/#details |



No comments:
Post a Comment