Any content you receive is for information purposes only. Always conduct your own research.*Sponsored
Krypton Street Just Put (Nasdaq: NXL) On Tomorrow's Radar
Don't Miss The Next Breakout—Get Real-Time Alerts Sent Directly
To Your Phone. Up To 10X Faster Than Email.
Here's Why We'll Be Watching (NXL) Before Tomorrow's
Session—February 24, 2026…
February 23, 2026
Tomorrow's Coverage | Pull Up (NXL) Tonight—It Just Hit Tuesday's Watchlist
Dear Reader, At Krypton Street, we keep our focus on early setups where the narrative is sharpening and the market is starting to pay attention—and Nexalin Technology, Inc. (Nasdaq: NXL) just stepped into that window. For many neurological and mental health conditions, today's standard care can reduce symptoms, yet a meaningful share of patients still don't get durable relief. (NXL) is working to address that gap with its proprietary Deep Intracranial Frequency Stimulation (DIFS™) platform—a non-invasive approach designed to influence key brain systems tied to mood, sleep, and cognitive function. That's just one of the reasons (NXL) is topping our watchlist tomorrow morning—Tuesday, February 24, 2026.
But keep in mind, (NXL) has less than 15M shares listed as available to the public, according to MarketWatch. When companies have small floats like this, the potential exists for big moves if demand begins to shift. And that's exactly why (NXL) has our attention right now. (NXL) is still sitting below $1 and appears to be flying under the radar of many screens—yet the setup is starting to change. Recently, (NXL) made an approximate 37% move inside of 3 days, from around $0.37 on February 17 to $0.51 on February 20, according to data available from Barchart. Moves like that don't happen in a vacuum—they often show up when a chart is waking up and supply-demand dynamics begin to tighten. The move pushed (NXL) above its 5-day moving average of around $0.44 and brought it within striking distance of its 20-day MA near $0.53. And with the 50-day and 100-day still higher—around $0.65 and $0.93, respectively—we're watching closely to see whether potential momentum continues to build from here. Even so, this isn't a name that's invisible across the board. At least one firm has stepped in with published research—and put a clear target on (NXL) from here. Coverage Update: $4 Target Suggests 700% Upside Potential
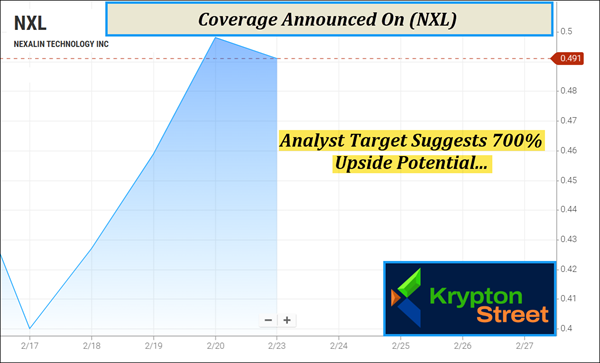
Analysts are beginning to recognize the potential for a significant valuation rerating. Anthony Vendetti of Maxim Group has issued a target of $4 for (Nasdaq: NXL). Based on the recent range of approximately $0.50, this suggests an upside potential of 700%. This target reflects the analyst's confidence in Nexalin's clinical progress, its growing patent portfolio, and its ability to execute on its global distribution strategy. When you combine this massive upside potential with the company's recent FDA milestones and international approvals, a clear thesis begins to emerge. (Nasdaq: NXL) is a company that is fundamentally stronger than its current price suggests, with multiple near-term catalysts that could drive increased market awareness. A New Frontier in Neurostimulation
Nexalin Technology, Inc. (Nasdaq: NXL) is a medical technology company developing neurostimulation products built around its patented Deep Intracranial Frequency Stimulation (DIFS™) platform. Unlike many approaches that primarily affect the brain's surface, Nexalin's proprietary waveform is designed to reach deeper mid-brain structures—areas tied to mood, sleep, and cognitive function. The company is transitioning from its Generation 1 device, which is FDA-cleared for anxiety, depression, and insomnia, to its more powerful Gen-2 SYNC console. This 15 mA system is positioned as the next stage of the platform, targeting larger indications such as Alzheimer's disease, chronic pain, and ADHD, while maintaining a non-invasive, dr-ug-free approach. Large and Growing $573B Market Backdrop

The global market for mental health and neurodegenerative care is evolving quickly. With the neuromodulation market forecasted to reach $11B by 2028, (NXL) is operating in an expanding category, while broader projections size the global mental health market at $573B by 2033. Nexalin is focused on conditions that have historically been difficult to treat. The global Alzheimer's disease market is projected to grow over 330%, from around $7.79B in 2026 to $33.62B by 2034, and the company's recent FDA Q-Submission for its Gen-2 SYNC device in Alzheimer's represents a meaningful step toward pursuing this expanding, underserved area. Beyond neurodegeneration, (NXL) is also advancing its work in ADHD, which affects approximately 4–5% of adults globally. A recent peer-reviewed study in Molecular Psychiatry reported that the technology could objectively normalize brain activity in these patients, positioning the company to pursue a share of the $10B+ ADHD treatment market. 
Recent Operational Achievements and Global Expansion
The last several months have been a period of rapid execution for (NXL). One of the most critical milestones was the regulatory approval and subsequent launch of its Gen-2 SYNC device in Israel. To facilitate this, the company appointed Carmi Masha Technologies Ltd. as its exclusive distributor in the region. This partnership is expected to accelerate commercial activities and provide real-world data to support Nexalin's broader international expansion. In the United States, (NXL) is moving toward pivotal clinical trials. In December 2025, the company successfully completed a substantive Q-Submission meeting with the FDA. This meeting provided clarity on the clinical development plan for the Gen-2 SYNC console in Alzheimer's disease. Alignment was reached on foundational components such as patient population and study endpoints, paving the way for a potential Investigational Device Exemption (IDE) filing. This disciplined regulatory strategy ensures that the company remains on a clear path toward potential U.S. authorization. Management Team and Scientific Validation

A company's technology is only as strong as the team behind it. (NXL) is led by: - Mark White — President & CEO
- Dr. David Owens — Chief Medical Officer
- John Patrick Claude — Co-Founder & Director of Engineering
The company also highlights a Military & Government Advisory Board that includes:
- General Wesley Clark — former NATO Supreme Allied Commander
- William A. Hudson, Jr. — former General Counsel, U.S. Department of Veterans Affairs
Together, this group brings deep experience across medical devices and clinical research, supporting (NXL)'s focus on scientific rigor. That research emphasis is reflected in the company's expanding neuroimaging work—using tools such as fMRI and MEG to evaluate how DIFS™ may influence brain network activity—helping differentiate Nexalin within the neuromodulation category. See the company's full corporate presentation here. Pipeline and Future Potential Catalysts
(NXL)'s roadmap extends beyond its current consoles. The company is developing the Gen-3 HALO™ headset for use in a virtual clinic setting—an at-home model designed to expand access while maintaining physician oversight. On February 5, 2026, Nexalin announced the launch of NeuroCare®, an AI-driven virtual clinic platform developed in collaboration with the University of California, San Diego (UCSD). NeuroCare is designed to support remote, physician-monitored neurotherapy using the Gen-3 HALO™ across Alzheimer's/dementia, mood disorders, TBI, and PTSD, while operating within a subscription-based model that management says can support recurring revenue and long-term patient engagement. The launch builds on Nexalin's ongoing clinical relationship with UCSD, where patient enrollment has begun in trials evaluating Deep Intracranial Frequency Stimulation, and management says the broader digital ecosystem is intended to capture clinical data and scale over time. In recent February presentations, CEO Mark White said the company is moving from development into execution, with Gen-3 HALO advancing into manufacturing and an initial production run underway. White also said Nexalin has aligned with the FDA on trial designs for insomnia and Alzheimer's/dementia, with recruitment expected in the coming months and early clinical data anticipated later this year. He framed the company's focus as "military, mood, and memory," highlighting UCSD as a key partner and pointing to the next 12–18 months as a window for FDA trial launches, continued data updates, and potential regulatory submissions tied to these core indications. Furthermore, (NXL) is exploring additional indications. A peer-reviewed case report in The American Journal on Addictions discussed the potential of the 15 mA device in treating an impulse-control disorder alongside alcohol-use comorbidity, reporting complete abstinence and improved cognitive control after 20 sessions—underscoring the platform's broader versatility. Here's 7 Reasons Why (NXL) Will Be Topping Our Watchlist Tomorrow
Morning Tuesday, February 24, 2026…
1. Recent Momentum: An approximate 37% move in just three days recently pushed (NXL) from about $0.37 to $0.51, signaling renewed activity on the chart.
2. Small Float: With fewer than 15M shares available to the public, (NXL)'s small float could have the potential for big moves if demand begins to shift. . 3. Technical Shift: The latest move carried (NXL) above its 5-day moving average and toward its 20-day level, while longer-term averages at $0.65 and $0.93 remain overhead. 4. FDA Alignment: Following a substantive Q-Submission meeting with the FDA, (NXL) has aligned on trial designs for Alzheimer's and insomnia, outlining a defined regulatory path. 5. Massive Markets: Operating within a neuromodulation market projected at $11B by 2028 and a broader mental health market estimated at $573B by 2033, (NXL) is positioned inside large and expanding categories. 6. Digital Expansion: The launch of NeuroCare® and development of the Gen-3 HALO™ headset signal that (NXL) is transitioning from development into execution with a home-based, physician-monitored platform. 7. Analyst Coverage: A published $4 target from Maxim Group implies about 700% upside potential from the recent $0.50 range, putting (NXL) on the radar of at least one firm following its clinical and regulatory progress. Pull Up (NXL) Before Tomorrow Morning…
When you line it all up, it's easy to see why (NXL) is back on the radar. The public float is tight (fewer than 15M shares), recent momentum has been decisive (about a 37% move in three days), and the chart is beginning to reflect that shift with (NXL) back above its 5-day moving average and working toward the 20-day level. At the same time, at least one covering firm has published a $4 target—implying about 700% upside potential from the recent $0.50 range—while the company continues to outline a clearer regulatory path after a substantive FDA Q-Submission process tied to Alzheimer's and insomnia. Layer in the larger backdrop—$11B projected for neuromodulation by 2028 and $573B projected for the broader mental health market by 2033—and the story starts to look less like a random ticker and more like a name worth understanding. Add the NeuroCare® launch and Gen-3 HALO™ development, and it is clear management is pushing toward a home-based, physician-monitored model designed to scale. We'll be tracking (NXL) closely at first light tomorrow. Pull up (NXL) tonight so you're not catching up in the morning. And keep an eye out for my morning update—it may hit earlier than usual. Have a good night. Sincerely, Alex Ramsay
Co-Founder / Managing Editor Krypton Street Newsletter
| 





No comments:
Post a Comment