| Latest Operational Successes + Worldwide Expansion
The last several months have been a period of rapid execution for NXL.
One of the most critical milestones was the regulatory approval and subsequent launch of its Gen-2 SYNC device in Israel.
To facilitate this, the company appointed Carmi Masha Technologies Ltd. as its exclusive distributor in the region.
This partnership is expected to accelerate commercial activities and provide real-world data to support Nexalin's broader international expansion.
In the United States, NXL is moving toward pivotal clinical trials.
In December 2025, the company successfully completed a substantive Q-Submission meeting with the FDA.
This meeting provided clarity on the clinical development plan for the Gen-2 SYNC console in Alzheimer’s disease.
Alignment was reached on foundational components such as patient population and study endpoints, paving the way for a potential Investigational Device Exemption (IDE) filing.
This disciplined regulatory strategy ensures that the company remains on a clear path toward potential U.S. authorization.
Sources & More Company Info: NXL Website. NXL Presentation. -----
(Nasdaq: NXL) And The 5 Potential Catalysts Leading The Way
1.) A Fairly Low Float Could Create The Environment For Explosive/Significant Volatility Potential.
With roughly 15.93Mn shares in its float, volatility potential could become heightened at the drop of a hat for NXL.
2.) An Analyst Target Suggests Potential Triple-Digit Upside From Current Chart Levels.
At this moment, an analyst target is suggesting strong potential upside for NXL.
A Maxim Group analyst target of $4.00 highlights a potential 650+% upside from Monday's close.
Could this target be suggesting NXL is undervalued from current chart levels?
3.) Nexalin Launches NeuroCare®, an AI-Driven Virtual Clinic Designed to Transform Access for Patients with Alzheimer’s, Mood Disorders, TBI & PTSD Care.
Nexalin Technology, Inc. has launched NeuroCare®, an AI-powered virtual clinic developed with UC San Diego to improve access and affordability for patients with Alzheimer’s, mood disorders, TBI, and PTSD.
The platform integrates Nexalin’s Gen‑3 HALO™ headset for remote, physician-guided treatments, creating a full digital heal-th ecosystem.
This combination enables the potential for recurring revenue through therapy subscriptions and continuous clinical data capture.
By pairing advanced neurostimulation with digital care delivery, Nexalin positions itself as a potential market leader in accessible, stigma-free mental and neurological heal-th solutions worldwide.
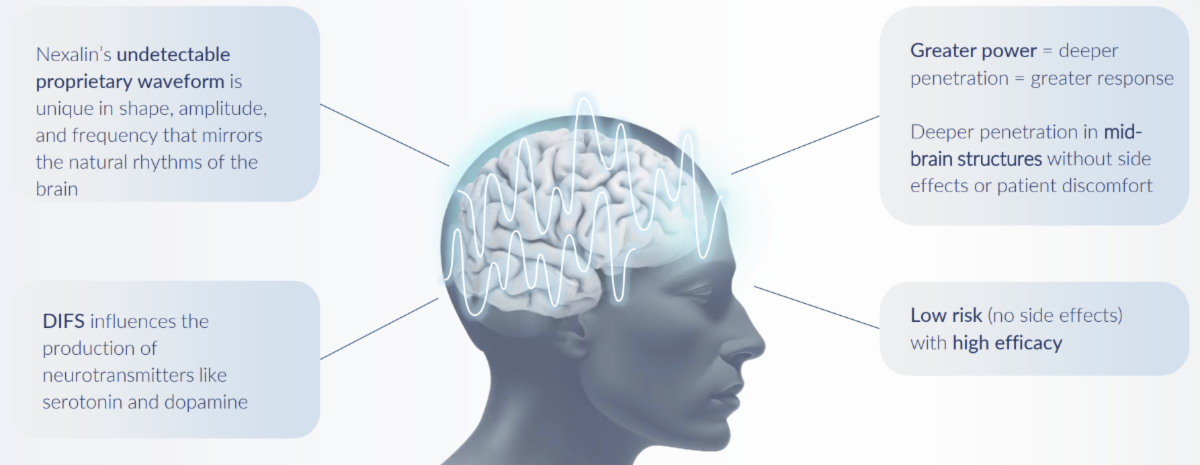
4.) Nexalin Highlights its Expanding Body of Peer-Reviewed Neuroimaging Research Confirming its DIFS™ Technology as the Leader in Evidenced-Based Non-Invasive Brain Stimulation.
Nexalin Technology’s ongoing peer-reviewed studies continue to validate its Deep Intracranial Frequency Stimulation (DIFS™) technology as the most advanced non-invasive brain therapy available.
Clinical imaging and data across conditions such as insomnia, depression, PTSD, TBI, Alzheimer’s, and ADHD demonstrate measurable normalization of neural function.
This data-driven evidence supports Nexalin’s FDA initiatives and reinforces the company’s “mood‑military‑memory” clinical strategy.
With proven ability to modulate deep brain networks, Nexalin’s results surpass traditional stimulators, strengthening its reputation as a science-backed pioneer in advanced neurostimulation therapies for mental and cognitive disorders.
5.) Nexalin Technology Completes FDA Q-Submission Meeting for Gen-2 SYNC™ Neurostimulation Console in Alzheimer’s Disease Program.
Nexalin Technology successfully completed a Q‑Submission meeting with the FDA for its Gen‑2 SYNC™ neurostimulation console targeting Alzheimer’s disease.
The agency’s encouraging feedback clarifies the regulatory roadmap, supporting a De Novo classification pathway.
Nexalin aligned with the FDA on pilot and pivotal study parameters, including patient selection, cognitive endpoints, and imaging metrics.
This regulatory milestone provides confidence in Nexalin’s strategy to advance safe, non-invasive, drug-free treatment alternatives.
The strong engagement positions Nexalin to accelerate U.S. trials and move closer to delivering breakthrough neurostimulation innovations addressing unmet neurological needs. -----
Now underway: coverage is kicked-off on Nexalin Technology, Inc. (Nasdaq: NXL).
Be on watch for updates coming out soon. Talk shortly.
All the best, Dane James Editor Market Pulse Today
(Remember: St-ock Prices Could Be Significantly Lower Now From The Original Dates I Provided.)
*MarketPulseToday.com (“MarketPulseToday” or “MPT” ) is owned by Thousand Sun Media LLC, MPT is not responsible for its accuracy. Make sure to always do your own research and due diligence on any day and swing profile MPT brings to your attention. Any emojis used do not have a specific defined meaning, and may be used inconsistently. We do not provide personalized in-vest-ment advice, are not in-vest-ment advisors, and any profiles we mention are not suitable for all in-vest-ors.
Pursuant to an agreement between Thousand Sun Media LLC and TD Media LLC, Thousand Sun Media LLC has been hired for a period beginning on 02/23/2026 and ending on 02/24/2026 to publicly disseminate information about (NXL:US) via digital communications. Under this agreement, TD Media LLC has paid Thousand Sun Media LLC seven thousand five hundred USD ("Funds"). These Funds were part of the seventy seven thousand five hundred USD funds that TD Media LLC received from a third party named Goldwyn Media LLC who did receive the Funds directly or indirectly from the Issuer and does not own st-ock in the Issuer but the reader should assume that the clients of the third party own shares in the Issuer, which they will liquidate at or near the time you receive this communication and has the potential to hurt share prices.
Neither Thousand Sun Media LLC, TD Media LLC and their member own shares of (NXL:US).
Please see important disclosure information here: https://marketpulsetoday.com/disclosure/nxl-2uade/#details | 

No comments:
Post a Comment